Category: Medications - Page 3

Biosimilars vs Generics: Understanding Authorized Biologic Alternatives
Biosimilars and authorized generics both lower drug costs, but they're not the same. Learn how FDA approval processes, manufacturing differences, and market adoption vary between these two drug types.
Feb, 5 2026

Dangerous Medical Abbreviations to Avoid: A Complete Guide to Prescription Safety
Learn about dangerous medical abbreviations that cause prescription errors and how healthcare providers prevent them. Essential for patients and professionals.
Feb, 4 2026

Levothyroxine with Iron and Calcium: How to Separate Doses for Maximum Effectiveness
Levothyroxine can lose effectiveness if taken with iron or calcium supplements. Learn how to space doses by at least 4 hours to ensure proper absorption and avoid worsening hypothyroid symptoms.
Feb, 1 2026

Drug Withdrawals and Recalls: Why Medications Get Removed from the Market
Medications are pulled from the market when new evidence shows they're unsafe or ineffective. Learn how the FDA decides to withdraw drugs, why it takes so long, and what patients can do to stay protected.
Jan, 29 2026

Therapeutic Equivalence: What It Really Means for Patient Safety
Therapeutic equivalence ensures generic drugs work just like brand-name ones, protecting patient safety while lowering costs. Learn how the FDA verifies this, what the Orange Book means, and why it matters for your health.
Jan, 28 2026

Spanish-Language Resources for Understanding Generic Medications
Spanish-language resources help patients understand that generic medications are just as effective as brand-name drugs. Learn where to find trusted guides, how to explain appearance differences, and why regional terminology matters for safe use.
Jan, 27 2026

Azathioprine and TPMT Testing: How Genetic Screening Prevents Life-Threatening Side Effects
Azathioprine is an effective but risky immunosuppressant. TPMT and NUDT15 genetic testing can prevent life-threatening blood cell crashes before they happen-especially for those with inherited enzyme deficiencies.
Jan, 26 2026
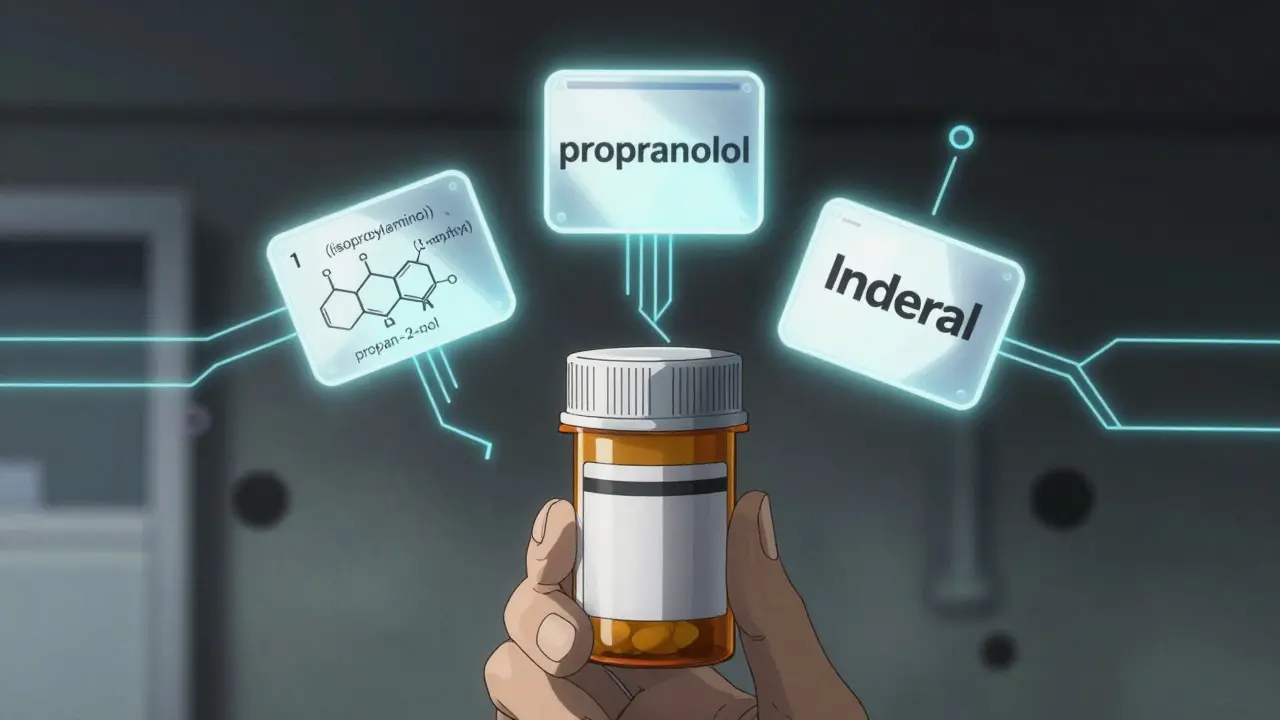
Drug Nomenclature: Chemical, Generic, and Brand Names Explained
Learn how drugs get their chemical, generic, and brand names - and why this system prevents dangerous medication errors. Understand the science behind the names on your prescriptions.
Jan, 26 2026

Generic Drug Classifications: Types and Categories Explained
Understand how generic drugs are classified by therapeutic use, mechanism, legal status, cost tiers, and naming conventions. Learn why these systems matter for your health, safety, and out-of-pocket costs.
Jan, 21 2026

Interchangeability: When Biosimilars Can Be Substituted Automatically in the U.S.
Interchangeable biosimilars can be swapped automatically at pharmacies in the U.S.-but only if FDA-approved and allowed by state law. Learn how they differ from generics, which ones are approved, and why patient and pharmacist confusion still runs high.
Jan, 20 2026
